Hard skills include abilities like sermon preparation, theological research, and conflict resolution that are essential for pastors to lead and support their congregations.
Popular Pastor Resume Examples
Discover our top pastor resume examples that emphasize key skills such as community outreach, sermon preparation, and leadership. These examples illustrate how to communicate your experiences and dedication to your faith.
Ready to build a remarkable resume? Our Resume Builder offers user-friendly templates specifically designed for religious leaders, helping you present your qualifications with confidence.
Entry-level pastor resume
This entry-level resume highlights the applicant's leadership abilities and community engagement achievements, showcasing a strong track record of increasing participation within the church. New professionals in ministry should illustrate their capacity for spiritual guidance and community outreach through measurable impacts, even when work experience is limited.
Mid-career pastor resume
This resume showcases a dynamic pastor with extensive leadership experience, emphasizing community outreach and ministry growth. The progression from youth minister to lead pastor demonstrates a robust capability for taking on greater responsibilities and fostering effective change within the congregation.
Experienced pastor resume
The work history section highlights the applicant's powerful pastoral roles, showcasing a 20% growth in congregation size and a 30% increase in outreach participation. The clear bullet points improve readability, making it an effective presentation for hiring committees seeking proven leadership.
Resume Template—Easy to Copy & Paste
Linda Martinez
Milwaukee, WI 53202
(555)555-5555
Linda.Martinez@example.com
Professional Summary
Passionate Pastor with 9 years of experience leading congregations, enhancing community engagement, and fostering spiritual growth. Skilled in public speaking, counseling, and event planning. Dedicated to building strong, supportive communities.
Work History
Pastor
Grace Community Church - Milwaukee, WI
March 2021 - March 2026
- Led a congregation of 500+ members.
- Increased participation in community events by 40%.
- Raised ,000 for outreach programs.
Associate Pastor
Central Baptist Church - Brookfield, WI
March 2016 - February 2021
- Prepared and delivered weekly sermons.
- Expanded youth ministry by 25%.
- Mentored new members and volunteers.
Youth Pastor
Faith Methodist Church - Waukesha, WI
March 2013 - February 2016
- Coordinated youth activities and programs.
- Increased teen membership by 35%.
- Organized annual youth retreats.
Skills
- Public speaking
- Leadership
- Community outreach
- Counseling
- Religious education
- Fundraising
- Event planning
- Youth ministry
Certifications
- Certified Pastoral Counselor - American Association of Pastoral Counselors
- Youth Ministry Certification - National Association of Youth Ministry
Education
Master of Divinity Theology
Vanderbilt University Nashville, Tennessee
May 2013
Bachelor of Arts Religious Studies
University of Tennessee Knoxville, Tennessee
May 2010
Languages
- Spanish - Beginner (A1)
- French - Intermediate (B1)
- German - Beginner (A1)
How to Write a Pastor Resume Summary
Your resume summary is the first thing employers will see, making it essential to create a lasting impression that reflects your qualifications as a pastor. In this role, you should highlight your experience in community engagement, spiritual guidance, and leadership skills to connect with potential congregants. To better understand what makes an effective pastor resume summary, consider the following examples that illustrate both successful and ineffective approaches:
I am a dedicated pastor with years of experience in ministry and community service. I seek a position where I can use my talents to help others and grow spiritually. My ideal church would support personal development and foster strong relationships within the congregation. I believe my presence could greatly benefit the community if given the chance.
- Lacks specific achievements or unique skills that showcase the applicant’s value.
- Relies heavily on personal desires rather than what they can contribute to the church or community.
- Uses vague language that does not highlight concrete experiences or results.
Compassionate pastor with over 10 years of experience in community outreach and spiritual guidance. Successfully increased church membership by 30% through innovative programs and engaging sermons that resonate with diverse audiences. Proficient in conflict resolution, pastoral care, and leading community service initiatives to foster a supportive environment.
- Begins with clear experience duration and areas of expertise relevant to pastoral duties.
- Highlights a quantifiable achievement that showcases effectiveness in community engagement.
- Mentions specific skills that demonstrate competencies necessary for effective pastoral leadership.
Pro Tip
Showcasing Your Work Experience
The work experience section is important for your resume as a pastor, serving as the primary area where you’ll present your qualifications. Effective resume templates always feature this section prominently.
This area should be organized in reverse-chronological order, detailing your previous roles within ministry or church leadership. Include three to four bullet points that highlight your accomplishments and contributions in each position.
To further assist you, we have prepared examples that showcase effective work history entries for pastors. These examples will illustrate what works well and what may not resonate with hiring committees:
Pastor
Community Church – Springfield, IL
- Led Sunday services.
- Provided counseling to church members.
- Organized community events.
- Worked with volunteers on various projects.
- Lacks specific details about the impact of services led.
- Bullet points are too vague and do not highlight unique contributions.
- Focuses on general responsibilities rather than measurable outcomes or achievements.
Pastor
Faith Community Church – Springfield, IL
March 2018 - Current
- Lead weekly worship services attended by over 200 congregants, fostering spiritual growth through engaging sermons.
- Develop and oversee community outreach programs that have increased participation by 40% in the last year.
- Provide pastoral care and counseling to individuals and families, contributing to a supportive church environment.
- Starts each bullet with strong action verbs that highlight the pastor’s contributions.
- Incorporates specific metrics reflecting the growth of community engagement and attendance.
- Highlights essential skills such as leadership, communication, and empathy relevant to pastoral duties.
While your resume summary and work experience often take center stage, it's essential not to overlook the other critical sections that contribute to a well-rounded document. For more detailed guidance on perfecting every part of your resume, be sure to consult our comprehensive guide on how to write a resume.
Top Skills to Include on Your Resume
A skills section is a vital component of any effective resume, offering job seekers the opportunity to highlight their strengths and capabilities. It serves as a quick reference for employers to gauge essential qualifications at a glance.
This section enables hiring managers to swiftly evaluate whether job seekers meet the criteria for the role, streamlining their selection process. For pastor professionals, it’s important to showcase both hard and soft skills relevant to ministry work, which will be discussed in detail below.
Soft skills like empathy, active listening, and humility are important for pastors to build trusting relationships and provide meaningful spiritual guidance.
When selecting skills for your resume, it’s important to align them with what employers anticipate from ideal applicants. Many organizations rely on automated screening systems that filter out applicants lacking essential resume skills.
To improve your chances, carefully review job postings for insights into the specific skills recruiters seek. By prioritizing these key competencies in your resume, you can improve your visibility to both hiring managers and ATS alike.
Pro Tip
10 skills that appear on successful pastor resumes
Improve your resume to attract recruiters by highlighting essential skills that are most valued in pastoral roles. You can find examples of these skills showcased in our resume examples, which will help you present yourself confidently to potential employers.
Here are 10 skills to consider adding to your resume if they align with your experiences and the job specifications:
Empathy
Public speaking
Counseling
Conflict resolution
Community outreach
Leadership
Crisis management
Interpersonal communication
Organizational abilities
Cultural competence
Based on analysis of 5,000+ social services professional resumes from 2023-2024
Resume Format Examples
Choosing the right resume format is important for pastors, as it highlights their spiritual leadership, community impact, and relevant experiences in a clear and compelling way.
Functional
Focuses on skills rather than previous jobs.
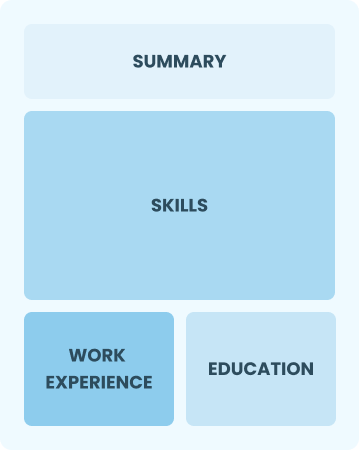
Best for:
Recent graduates and career changers with up to two years of experience.
Combination
Balances skills and work history equally.
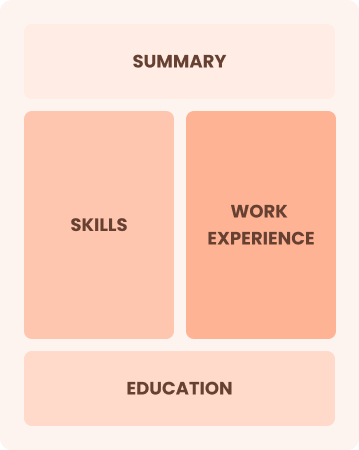
Best for:
Mid-career professionals focused on demonstrating their skills and seeking growth opportunities.
Chronological
Emphasizes work history in reverse order.

Best for:
Seasoned pastors leading dynamic congregational growth and community outreach.
Frequently Asked Questions
Should I include a cover letter with my pastor resume?
Absolutely. Including a cover letter is essential as it highlights your unique qualifications and shows your enthusiasm for the position. It gives you a chance to connect personally with potential employers. If you're looking for guidance, explore our resources on how to write a cover letter or use our Cover Letter Generator to streamline the process and create one that impresses.
Can I use a resume if I’m applying internationally, or do I need a CV?
When you're applying for international positions, use a CV instead of a resume, as it offers a comprehensive overview of your academic and professional history. Explore our how to write a CV resources and CV examples to ensure your application meets the expectations of employers worldwide.
What soft skills are important for pastors?
Soft skills such as interpersonal skills like compassion, active listening, and conflict resolution are important for a pastor. These abilities foster meaningful connections with congregants and facilitate effective community engagement, ensuring a supportive environment where individuals feel valued and understood.
I’m transitioning from another field. How should I highlight my experience?
Highlight your transferable skills such as communication, empathy, and community engagement when applying for pastor positions. These abilities showcase your potential to connect with and guide congregants, even if your experience in ministry is limited. Provide concrete examples from past roles that illustrate how you can fulfill the responsibilities of a pastor.
Where can I find inspiration for writing my cover letter as a pastor?
If you're seeking inspiration for your cover letter, check out our expertly crafted cover letter examples tailored for pastor positions. These samples provide valuable insights into effective content presentation, formatting tips, and ways to showcase your unique qualifications. Let them guide you in crafting a compelling application.
How do I write a resume with no experience?
Crafting a resume with no experience for pastor positions can still showcase your strengths. Highlight volunteer work, community involvement, and any leadership roles you've taken on. Focus on your passion for guiding others and your ability to connect with diverse groups. Employers value dedication and heart, so let that shine through.







