Hard skills cover precise measurement techniques, quality assurance methodologies, and expertise in using inspection tools to ensure products meet industry standards for quality control inspectors.
Popular Quality Control Inspector Resume Examples
Check out our top quality control inspector resume examples that emphasize important skills such as attention to detail, problem-solving, and adherence to safety standards. These examples will help you showcase your expertise to potential employers.
Ready to build a standout resume? Our Resume Builder offers user-friendly templates specifically designed for quality control professionals, making the process simple and efficient.
Recommended
Entry-level quality control inspector resume
This entry-level resume for a quality control inspector highlights the applicant's achievements in process optimization and compliance, showcasing significant contributions to previous employers. New professionals must convey their problem-solving skills, relevant certifications, and powerful accomplishments to attract potential employers despite having limited professional experience.
Mid-career quality control inspector resume
This resume effectively showcases the applicant's extensive experience in quality control, highlighting achievements in process optimization and compliance. The progression from specialist to inspector indicates strong leadership potential and readiness for more complex challenges within quality management.
Experienced quality control inspector resume
This resume highlights the applicant's extensive experience as a Quality Control Inspector, showcasing significant achievements such as a 25% reduction in rework and a perfect audit success rate. The use of bullet points improves clarity and allows potential employers to quickly identify key accomplishments.
Resume Template—Easy to Copy & Paste
Chris Jones
Riverside, CA 92508
(555)555-5555
Chris.Jones@example.com
Skills
- Statistical Analysis
- Quality Assurance
- ISO Standards Compliance
- Process Improvement
- Defect Reduction Strategies
- Quality Audits
- Data Interpretation
- Continuous Improvement
Certifications
- Certified Quality Inspector (CQI) - American Society for Quality
- Six Sigma Green Belt - International Association for Six Sigma Certification
Languages
- Spanish - Beginner (A1)
- Mandarin - Beginner (A1)
- German - Beginner (A1)
Professional Summary
Accomplished Quality Control Inspector with expertise in ISO standards and defect reduction strategies. Proven track record in enhancing compliance and boosting process efficiency with statistical analysis.
Work History
Quality Control Inspector
Precision Manufacturing Group - Riverside, CA
March 2022 - March 2026
- Reduced defects by 25% in 15-month period
- Implemented ISO 9001 standards across production floor
- Carried out weekly audits, improving compliance by 30%
Quality Assurance Analyst
Innovate Engineering Solutions - San Diego, CA
June 2019 - February 2022
- Supervised quality control measures, reducing errors by 18%
- Standardized testing procedures, boosting consistency by 40%
- Analyzed reports to enhance quality measures with 15% efficiency
Product Quality Evaluator
TechVision Components Inc. - Riverside, CA
March 2016 - May 2019
- Tested product durability, reducing return rates by 10%
- Developed inspection protocol, resulting in 22% productivity gain
- Ensured compliance with industry standards, improving safety
Education
Master of Science Quality Management
University of Washington Seattle, Washington
June 2016
Bachelor of Science Industrial Engineering
Washington State University Pullman, Washington
June 2014
How to Write a Quality Control Inspector Resume Summary
Your resume summary is the first opportunity to make a lasting impression on hiring managers. This critical section should effectively showcase your skills and relevant experience as a quality control inspector. Highlight your attention to detail, analytical abilities, and commitment to maintaining high standards in production. To see what makes an powerful summary, review these quality control inspector resume summary examples that will clarify effective strategies:
I am a dedicated quality control inspector with many years of experience in the field. I am seeking a position where I can use my skills and help the company achieve its goals. A role that offers good work-life balance and opportunities for advancement is what I am looking for. I believe my background will be beneficial to your team.
- Contains general statements about experience without providing specific achievements or skills
- Emphasizes the applicant's needs over what they can contribute to the employer, which is less appealing
- Uses vague language such as 'many years of experience' instead of quantifying expertise or demonstrating value
Detail-oriented quality control inspector with over 7 years of experience in manufacturing environments, specializing in product inspections and compliance auditing. Improved product quality by reducing defects by 20% through implementing effective inspection protocols and training staff on best practices. Proficient in ISO standards, statistical process control (SPC), and using various measurement tools to ensure adherence to specifications.
- Begins with a clear statement of experience level and area of expertise
- Highlights a quantifiable achievement that indicates direct impact on product quality
- Mentions relevant technical skills that are important for success in quality control roles
Pro Tip
Showcasing Your Work Experience
The work experience section is important for your resume as a quality control inspector, serving as the primary focus where you will present the bulk of your content. Good resume templates always ensure this section is prominent.
This area should be organized in reverse-chronological order, detailing your previous positions. Use bullet points to communicate your key achievements and responsibilities in each role.
To help you understand what makes an impact, we’ve provided a couple of examples showcasing effective work history entries for quality control inspectors. These examples illustrate what works well and what to avoid:
Quality Control Inspector
ABC Manufacturing – Dallas, TX
- Inspected products for quality.
- Filled out reports and checked documents.
- Collaborated with production teams.
- Ensured safety standards were met.
- Lacks specific details about the types of products inspected
- Bullet points do not highlight any achievements or improvements made
- Focuses on basic duties instead of measurable contributions to quality control
Quality Control Inspector
ABC Manufacturing – Dallas, TX
March 2020 - Current
- Conduct thorough inspections of incoming materials and finished products, ensuring compliance with industry standards and specifications.
- Implement quality control procedures that reduced product defects by 30%, improving overall production efficiency.
- Train new team members on inspection processes and safety protocols, fostering a culture of quality awareness throughout the facility.
- Starts each bullet point with dynamic action verbs to convey the applicant's contributions clearly
- Incorporates specific metrics to highlight significant achievements in quality improvement
- Showcases relevant skills such as training and procedure implementation, aligning well with the role's requirements
While your resume summary and work experience are important components, don't overlook the significance of other sections that can improve your application. Each part of your resume deserves careful attention to detail. For more in-depth tips, be sure to explore our comprehensive guide on how to write a resume.
Top Skills to Include on Your Resume
A skills section is vital for any resume, providing immediate clarity on your professional strengths. It enables job seekers to present their capabilities effectively and assists employers in identifying suitable job seekers swiftly.
This section allows hiring managers to quickly evaluate your match with the role's requirements. Quality control inspectors should highlight both technical and interpersonal skills, facilitating a deeper discussion of these attributes below.
Soft skills are essential for quality control inspectors as they improve communication, teamwork, and problem-solving abilities. These skills ensure effective collaboration and maintain high standards in product quality.
When selecting resume skills for your resume, it's best to align them with what employers expect. Many organizations use automated systems to screen out applicants lacking essential skills for the position.
Review job postings closely to improve your chances of getting noticed. They often highlight key skills that both recruiters and ATS prioritize, ensuring your application stands out in the selection process.
Pro Tip
10 skills that appear on successful quality control inspector resumes
Improving your resume with in-demand skills can significantly capture the interest of recruiters looking for quality control inspectors. You can observe how these skills are effectively highlighted in various resume examples, giving you the confidence to apply for positions successfully.
Consider including the following 10 skills in your resume if they align with your expertise and job expectations:
Attention to detail
Analytical thinking
Problem-solving
Communication skills
Team collaboration
Technical skill with inspection tools
Time management
Knowledge of quality standards
Report writing
Adaptability
Based on analysis of 5,000+ production professional resumes from 2023-2024
Resume Format Examples
Choosing the appropriate resume format is important for a quality control inspector, as it highlights your relevant skills and experiences while clearly outlining your career advancement in the field.
Functional
Focuses on skills rather than previous jobs
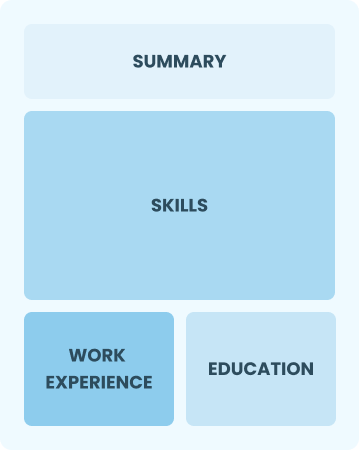
Best for:
Recent graduates and career changers with up to two years of experience
Combination
Balances skills and work history equally
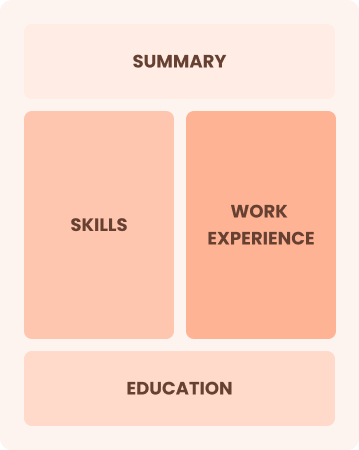
Best for:
Mid-career professionals focused on demonstrating their skills and advancing their careers
Chronological
Emphasizes work history in reverse order

Best for:
Experts leading quality assurance initiatives with proven track records
Frequently Asked Questions
Should I include a cover letter with my quality control inspector resume?
Absolutely, including a cover letter can significantly improve your application by showcasing your personality and passion for the job. It gives you a chance to highlight specific experiences that align with the role. For tips on crafting an effective cover letter, consider checking out our how to write a cover letter guide or using our Cover Letter Generator for quick assistance.
Can I use a resume if I’m applying internationally, or do I need a CV?
When applying for jobs abroad, a CV is often preferred over a resume. A CV provides a comprehensive overview of your academic and professional history. To help you craft an effective CV, explore our resources that offer formatting tips and CV examples tailored to international standards. For additional guidance on creating your document, learn how to write a CV.
What soft skills are important for quality control inspectors?
Soft skills like attention to detail, problem-solving, and effective communication are essential for quality control inspectors. These interpersonal skills foster collaboration with production teams and improve the ability to identify issues promptly, ensuring products meet high standards and maintaining customer trust.
I’m transitioning from another field. How should I highlight my experience?
Highlight your transferable skills like attention to detail, analytical thinking, and teamwork, which can apply to a quality control inspector role. Draw from previous experiences to showcase how these strengths helped you achieve success in past positions. Specific examples will illustrate your ability to maintain high standards and ensure product quality.
Should I use a cover letter template?
Yes, using a cover letter template tailored for quality control inspectors is advisable as it provides a clear structure and helps organize content well. This ensures you can prominently feature essential skills like attention to detail, knowledge of quality standards, and relevant certifications that resonate with hiring managers.
Should I include a personal mission statement on my quality control inspector resume?
Yes, including a personal mission statement in your resume is highly recommended. It conveys your core values and career aspirations. This approach is particularly effective when applying to companies that emphasize quality assurance and have a culture focused on continuous improvement.







