Hard skills are specific abilities like data analysis, problem-solving, and software expertise that associates should showcase to demonstrate their technical competence.
Popular Associate Resume Examples
Check out our top associate resume examples that emphasize skills such as teamwork, communication, and problem-solving. These samples are designed to help you showcase your achievements and appeal to potential employers.
Looking to create a standout resume? Our Resume Builder offers user-friendly templates tailored for associates, making the process simple and effective.
Recommended
Entry-level associate resume
This entry-level resume highlights the applicant's analytical skills and project management experiences gained through previous roles, showcasing achievements like significant ROI growth and improved operational efficiency. New professionals must demonstrate their ability to drive results and collaborate across teams through quantifiable accomplishments, even with limited work history.
Mid-career associate resume
This resume effectively presents the job seeker's extensive experience in client management and strategic planning, showcasing their readiness for leadership roles. The quantifiable achievements highlight a proven ability to improve service delivery and customer satisfaction, indicating strong capability for future challenges.
Experienced associate resume
The work history section highlights the applicant's strong capabilities in project management and client relations. Notable achievements include improving efficiency by 15% and boosting client satisfaction ratings by 20%, with a clear bullet point format improving readability for quick assessment of skills.
Resume Template—Easy to Copy & Paste
Jane Wu
Silverlake, WA 98290
(555)555-5555
jane@example.com
Professional Summary
Dynamic Associate with 6 years of experience in project management, data analytics, and client relations. Proven track record of enhancing productivity and increasing profitability. Strong strategic planning and leadership skills.
Work History
Associate
Insight Consulting Group - Silverlake, WA
April 2024 - April 2026
- Managed client accounts, increasing retention by 20%
- Developed strategies that boosted profit by 10%
- Coordinated projects, reducing delays by 15%
Project Coordinator
Visionary Solutions - Tacoma, WA
April 2021 - March 2024
- Supervised teams, enhancing productivity by 25%
- Led project meetings, decreasing issues by 18%
- Implemented tools, cutting costs by 12%
Junior Analyst
Innovative Analytics - Tacoma, WA
April 2020 - March 2021
- Assessed data sets, improving predictions by 15%
- Collaborated with senior staff, aiding in 5 projects
- Developed reports, increasing clarity by 30%
Skills
- Project Management
- Data Analysis
- Client Relations
- Strategic Planning
- Team Leadership
- Cost Reduction
- Time Management
- Analytics Tools
Certifications
- Certified Project Manager (CPM) - Project Management Institute
- Certified Data Analyst - Data Analytics Society
Education
January 2020
University of California, Berkeley Eastside, WA
Languages
- Spanish - C2
- Proficient - French
- B1 - Intermediate
- German - A2
- Elementary
How to Write an Associate Resume Summary
Your resume summary is the first thing employers will see, making it your chance to create a memorable first impression. For an associate role, it's essential to highlight relevant skills and experiences that showcase your ability to contribute effectively to the team. To guide you in crafting a powerful summary, here are some examples that illustrate what works well and what doesn't work:
I am an associate with experience in various roles and I am eager to find a position where I can apply my skills and contribute positively. My ideal job is one that offers personal development and a supportive environment. I believe that with the right opportunity, I can be a valuable part of any team.
- Uses vague language about experience without providing specific examples of skills or achievements
- Emphasizes personal desires over what value the job seeker brings to potential employers
- Lacks concrete details that demonstrate qualifications, making it less compelling
Compassionate nursing associate with 4 years of experience in patient care within hospital environments, focusing on assisting with daily living activities and vital signs monitoring. Improved patient satisfaction scores by 20% through effective communication and empathetic support. Proficient in electronic health records management, basic life support (BLS), and collaborating with healthcare teams to ensure quality care.
- Begins with a clear indication of experience level and primary focus area
- Highlights a quantifiable achievement that illustrates a positive impact on patient satisfaction
- Mentions relevant technical skills that align closely with the responsibilities of a nursing associate
Pro Tip
Showcasing Your Work Experience
The work experience section is the centerpiece of your resume as an associate. This is typically where you will find the bulk of your content, and good resume templates make sure to include this important part.
Organize this section in reverse-chronological order, detailing your previous positions. Use bullet points to highlight key achievements and responsibilities that showcase your skills and contributions in each role.
To illustrate effective work history entries for associates, we'll provide a couple of examples. These examples will clarify what works well and what pitfalls to avoid.
Sales Associate
Local Retail Store – Anytown, USA
- Helped customers find products.
- Assisted with checkout processes.
- Kept the store tidy and organized.
- Handled returns and exchanges.
- Lacks specific employment dates to indicate duration of experience
- Bullet points are vague and do not showcase achievements or skills
- Focuses on routine tasks instead of measurable contributions to sales or customer satisfaction
Sales Associate
Tech Gadgets Inc. – San Francisco, CA
March 2020 - Present
- Assist customers in selecting the right tech products by providing tailored recommendations, resulting in a 30% increase in customer satisfaction ratings.
- Achieve monthly sales goals consistently by using upselling techniques and fostering strong customer relationships.
- Train new staff on product knowledge and customer service best practices, improving team performance and cohesion.
- Starts each bullet with strong action verbs that clearly communicate the applicant’s contributions
- Incorporates specific metrics to illustrate the job seeker’s achievements effectively
- Highlights relevant skills and responsibilities that align with the job requirements
While your resume summary and work experience are important components, don’t overlook the importance of other sections that contribute to a well-rounded presentation. For more detailed guidance on crafting each part of your resume, be sure to explore how to write a resume for a comprehensive guide.
Top Skills to Include on Your Resume
A well-crafted skills section is a vital component of an effective resume, offering immediate clarity on your qualifications. It acts as a powerful tool for job seekers to highlight relevant capabilities while providing employers with a quick reference point for assessing job seeker potential.
This section enables hiring managers to swiftly evaluate whether applicants meet essential criteria and helps applicants directly connect their expertise to specific job demands. Associate professionals should emphasize both technical abilities and interpersonal skills, which will be detailed further below.
Soft skills include teamwork, adaptability, and problem-solving—important interpersonal abilities that help build positive relationships and boost productivity in an associate role.
When selecting skills for your resume, it’s important to align them with what employers expect from ideal applicants. Many organizations use automated systems to screen out applicants who lack the essential resume skills required for the position.
To improve your chances, carefully examine job postings for insights on which skills are most valued by recruiters and ATS. Highlighting these prioritized skills in your resume can significantly improve your visibility in the hiring process.
Pro Tip
10 skills that appear on successful associate resumes
Improving your resume with high-demand skills can attract the attention of recruiters looking for associates. Our resume examples highlight how these skills can be showcased, giving you the confidence to apply for roles that align with your qualifications.
Here are 10 essential skills you might want to feature on your resume if they align with your experience and the job requirements:
Team collaboration
Problem-solving
Customer service orientation
Time management
Adaptability
Data analysis
Attention to detail
Technical skill
Interpersonal communication
Project management
Based on analysis of 5,000+ administrative professional resumes from 2023-2024
Resume Format Examples
Choosing the right resume format is important as it highlights your most relevant qualifications and achievements, helping employers quickly identify your potential contributions to their organization.
Functional
Focuses on skills rather than previous jobs
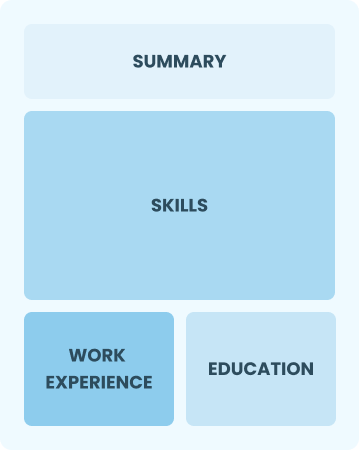
Best for:
Recent graduates and career changers with limited experience in the industry
Combination
Balances skills and work history equally
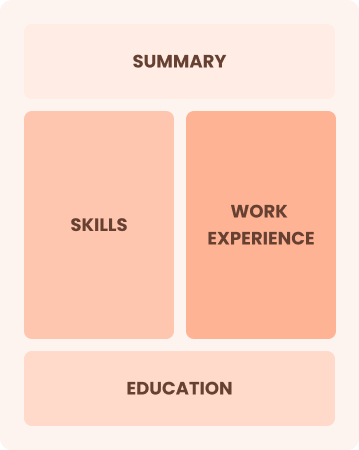
Best for:
Mid-career professionals focused on demonstrating their skills and pursuing growth opportunities
Chronological
Emphasizes work history in reverse order

Best for:
Seasoned professionals leading innovative teams
Frequently Asked Questions
Should I include a cover letter with my associate resume?
Absolutely, including a cover letter can significantly improve your application by showcasing your personality and specific qualifications for the role. It’s a chance to connect the dots between your resume and the job requirements. For assistance, explore our comprehensive guide on how to write a cover letter or use our Cover Letter Generator to craft one quickly and easily.
Can I use a resume if I’m applying internationally, or do I need a CV?
When applying for jobs outside the U.S., it's often better to use a CV instead of a resume. A CV provides a comprehensive overview of your academic and professional history. For guidance on how to write a CV and improve your international applications, explore our resources on effective formatting. Additionally, you can refer to these CV examples to see successful templates in action.
What soft skills are important for associates?
Soft skills such as communication, problem-solving, and interpersonal skills are important for associates. These abilities foster collaboration with team members and improve customer interactions, leading to a more productive and harmonious work environment where everyone feels valued and understood.
I’m transitioning from another field. How should I highlight my experience?
Highlight your transferable skills like communication, teamwork, and adaptability when applying for associate positions. These abilities show your potential to excel in the new role, even without direct experience. Share concrete examples from previous jobs where you applied these skills to demonstrate how they relate to an associate's responsibilities.
Where can I find inspiration for writing my cover letter as an associate?
If you’re pursuing an associate position, consider exploring cover letter examples. These samples offer valuable insights into content ideas and formatting that can help you showcase your qualifications while crafting a compelling narrative to engage potential employers.
Should I include a personal mission statement on my associate resume?
Yes, including a personal mission statement in your resume is advisable. It showcases your core values and career aspirations, making it particularly beneficial when targeting organizations that emphasize a strong cultural or ethical mission, helping you stand out as a job seeker aligned with their goals.








