Hard skills involve analytical abilities such as data analysis, testing methodologies, and familiarity with quality assurance tools that are important for quality analysts.
Popular Quality Analyst Resume Examples
Check out our top quality analyst resume examples that emphasize key skills such as data analysis, problem-solving, and process improvement. These examples demonstrate how to highlight your accomplishments in ways that catch the eye of potential employers.
Ready to build your ideal resume? The Resume Builder includes user-friendly templates tailored for quality professionals, making it simple to create a standout application.
Recommended
Entry-level quality analyst resume
This entry-level resume for a quality analyst highlights the job seeker's strong background in quality assurance, showcasing achievements like a 15% reduction in defect rates and improvements in testing efficiency. New professionals need to show their understanding of quality methodologies and their ability to contribute to process improvements, even with limited industry experience.
Mid-career quality analyst resume
This resume effectively showcases the job seeker's extensive experience and achievements in quality assurance, highlighting a trajectory of increasing responsibility. The focus on process improvement and team leadership illustrates readiness for more complex challenges, positioning them as a strong job seeker for advanced roles.
Experienced quality analyst resume
This work history section effectively highlights the applicant's extensive experience as a Quality Analyst, showcasing impressive achievements such as improving test efficiency by 30% and reducing defect rates by 40%. The clear formatting with bullet points improves readability, making it accessible for hiring managers to quickly identify key accomplishments.
Resume Template—Easy to Copy & Paste
Stephanie Butler
Portland, ME 04102
(555)555-5555
stephanie@example.com
Professional Summary
Experienced Quality Analyst skilled in QA and data analysis. Proven track record of enhancing product quality and optimizing processes. Strong background in testing automation and team leadership.
Work History
Quality Analyst
Precision QA Solutions - Portland, ME
May 2023 - April 2026
- Enhanced testing efficiency by 25%
- Reduced error rate by 30% via automation
- Led a team to deliver 12% faster results
Quality Assurance Specialist
Tech Verify Inc. - Portland, ME
March 2018 - April 2023
- Improved product quality by 20%
- Optimized processes reducing costs 15%
- Implemented QA protocols increasing efficiency
Quality Control Analyst
Golden Gate Software - Portland, ME
April 2015 - February 2018
- Conducted 500+ tests improving systems
- Streamlined workflows boosting output 10%
- Analyzed data reducing issues by 18%
Languages
- Spanish - B1
- Intermediate - French
- C1 - Advanced
- German - B1
- Intermediate -
Skills
- Quality Assurance
- Testing Automation
- Process Optimization
- Data Analysis
- Team Leadership
- Problem Solving
- Project Management
- Software Testing
Certifications
- Certified Quality Analyst - International QA Board
- Software Testing Professional - Tech Cert Institute
Education
Master of Science Computer Science
Stanford University Westbrook, ME
June 2014
Bachelor of Science Information Technology
University of California, Berkeley Westbrook, ME
June 2012
How to Write a Quality Analyst Resume Summary
Your resume summary is the first thing employers will read, making it important to create a compelling introduction. As a quality analyst, you should emphasize your attention to detail and analytical skills, showcasing how these qualities contribute to product excellence. The following examples will illustrate effective strategies for crafting your summary, helping you identify what elements resonate well and which may fall flat:
I am a dedicated quality analyst with years of experience in the field. I am seeking a position that allows me to use my skills and contribute positively to the team. A role with good benefits and opportunities for advancement would be perfect for me. I believe I can add value if given a chance.
- Contains broad statements about experience without providing specific achievements or skills
- Relies heavily on personal pronouns, making it feel less professional and focused
- Emphasizes job seeker's desires rather than highlighting how they can benefit the potential employer
Detail-oriented quality analyst with over 4 years of experience in software testing and process improvement. Achieved a 30% reduction in defect rates through the implementation of automated testing frameworks and improved QA methodologies. Proficient in SQL, JIRA, and Selenium, with a strong focus on delivering high-quality products that meet customer requirements.
- Starts with specific experience level and area of expertise
- Highlights quantifiable achievements that demonstrate measurable impact on quality assurance processes
- Lists relevant technical skills that are important for quality analyst roles
Pro Tip
Showcasing Your Work Experience
The work experience section is the centerpiece of your resume as a quality analyst, where you'll present the bulk of your content. Excellent resume templates always emphasize this important section.
This portion should be organized in reverse-chronological order, showcasing each of your past roles clearly. Use bullet points to highlight specific achievements and contributions in every position you've held.
Now, let's look at a couple of examples that will illustrate effective entries for a quality analyst's work history. These examples will help you identify what works well and what may not resonate as strongly:
Quality Analyst
Tech Solutions Inc. – San Francisco, CA
- Tested software for bugs.
- Documented issues and wrote reports.
- Collaborated with developers.
- Ensured quality standards were met.
- Lacks specific employment dates to establish timeline
- Bullet points are vague and do not highlight unique skills or achievements
- Focuses on routine tasks instead of measurable outcomes or contributions
Quality Analyst
Tech Solutions Inc. – San Francisco, CA
March 2020 - Current
- Conduct comprehensive testing of software applications, identifying and documenting over 150 bugs to improve product quality.
- Collaborate with development teams to streamline testing processes, resulting in a 30% reduction in time-to-release for major updates.
- Develop and implement automated testing scripts that increased test coverage by 40%, ensuring consistent performance and reliability.
- Uses strong action verbs to clearly articulate the applicant’s contributions and achievements
- Incorporates specific metrics that highlight the applicant’s effectiveness and impact on the organization
- Demonstrates relevant skills in software testing and quality assurance practices that are essential for the role
While your resume summary and work experience sections are important, don't overlook the importance of other areas that can make a significant impact. Each section deserves attention to detail. For more insights on crafting a standout resume, explore our how to write a resume guide.
Top Skills to Include on Your Resume
Including a skills section on your resume is important as it presents a clear snapshot of your qualifications to potential employers. This targeted approach helps job seekers stand out by aligning their capabilities with the needs of hiring managers.
For hiring professionals, this section offers an efficient way to gauge job seeker suitability at a glance, streamlining the selection process. Quality analyst job seekers should emphasize both technical skills and interpersonal abilities, which will be detailed further below.
Soft skills are essential for quality analysts because they foster effective communication, teamwork, and problem-solving abilities. This leads to improved project outcomes and greater client satisfaction.
Choosing resume skills that align with employer expectations and navigate automated screening systems is important. Many organizations use software to filter out applicants who lack essential skills for the position.
To make sure your resume stands out, review job postings carefully to gain insights into which skills are most valued by recruiters and applicant tracking systems (ATS). Highlighting these specific skills will improve your chances of passing through initial screenings and attracting the attention of hiring managers.
Pro Tip
10 skills that appear on successful quality analyst resumes
Highlighting key skills in your resume can greatly impact recruiters, particularly for quality analyst roles. You can see these essential skills incorporated in our resume examples, which will help you apply with confidence.
Consider including these 10 skills in your resume if they match your experience and job requirements:
Data analysis
Attention to detail
Problem-solving
Communication
Time management
Familiarity with QA methodologies
Software testing expertise
Project management
Risk assessment
Team collaboration
Based on analysis of 5,000+ information technology (it) professional resumes from 2023-2024
Resume Format Examples
Choosing the right resume format is important for a quality analyst. It highlights your analytical skills and relevant experience, making it easier for employers to recognize your career growth.
Functional
Focuses on skills rather than previous jobs
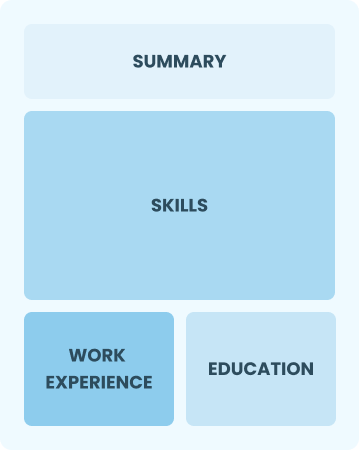
Best for:
Recent graduates and career changers with limited experience in quality analysis
Combination
Balances skills and work history equally
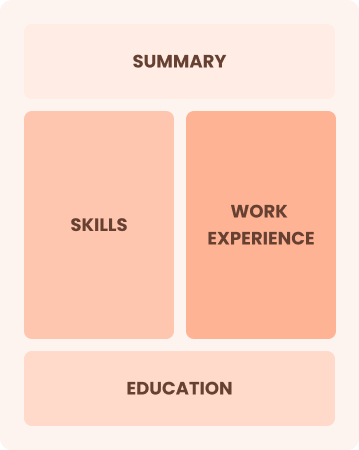
Best for:
Mid-career professionals focused on demonstrating their skills and growth potential
Chronological
Emphasizes work history in reverse order

Best for:
Seasoned analysts excelling in process improvement and team leadership
Frequently Asked Questions
Should I include a cover letter with my quality analyst resume?
Absolutely, including a cover letter is important for making a strong impression on potential employers. It allows you to highlight your relevant skills and show your enthusiasm for the position. If you're unsure how to craft one, consider exploring how to write a cover letter or using our Cover Letter Generator for a quick start.
Can I use a resume if I’m applying internationally, or do I need a CV?
When applying for jobs outside the U.S., a CV is often preferred over a resume. This is especially true in Europe and Asia. To create an effective CV, explore our resources on how to write a CV that aligns with international expectations. Additionally, review various CV examples to understand different formats and styles.
What soft skills are important for quality analysts?
Soft skills like attention to detail, problem-solving, and effective communication are important for quality analysts. These interpersonal skills foster collaboration with teams and improve the ability to understand user needs, ultimately leading to improved product quality and stronger working relationships.
I’m transitioning from another field. How should I highlight my experience?
Highlight your transferable skills such as attention to detail, analytical thinking, and effective communication from prior roles. These abilities show your potential to excel in quality analyst positions, even if your experience is outside the field. Use concrete examples to illustrate how your past successes align with key responsibilities in this role.
How should I format a cover letter for a quality analyst job?
When preparing to format a cover letter for quality analyst roles, start with your contact information and a respectful greeting. Use the opening paragraph to capture the reader’s interest by providing a concise summary of your skills. Emphasize how your experience matches the position's requirements and conclude with a compelling closing statement that encourages further conversation.
Should I include a personal mission statement on my quality analyst resume?
Yes, including a personal mission statement in your resume is recommended because it showcases your values and career aspirations. This approach works particularly well when applying to companies that prioritize cultural fit or maintain a strong focus on their core values.








