Technical abilities, known as hard skills, include project management, programming languages, and system analysis. These skills are important for a technical manager to oversee projects.
Popular Technical Manager Resume Examples
Discover our top technical manager resume examples that emphasize key skills like project management, team leadership, and strategic planning. These examples will help you showcase your achievements to potential employers.
Ready to design your ideal resume? Our Resume Builder offers customizable templates specifically crafted for technical professionals, making it easier than ever to highlight your expertise and experience.
Recommended
Entry-level technical manager resume
This entry-level resume for a technical manager highlights the job seeker's leadership abilities, project management skills, and technical accomplishments from previous roles. New professionals in this field must showcase their capacity to manage projects and lead teams, demonstrating relevant skills and certifications to attract potential employers despite limited experience.
Mid-career technical manager resume
This resume effectively showcases the applicant's extensive experience and quantifiable achievements, illustrating their capability for advanced technical leadership. The focus on project success, team mentorship, and strategic planning reflects a strong readiness to tackle complex challenges in evolving technological environments.
Experienced technical manager resume
The work history section highlights the applicant's strong leadership in technology management, showcasing achievements like a 23% increase in operational efficiency and $1.2M saved through budget optimization. The clear bullet point format improves readability, making it easy for hiring managers to quickly assess key accomplishments.
Resume Template—Easy to Copy & Paste
Suki Johnson
Springfield, IL 62709
(555)555-5555
Suki.Johnson@example.com
Professional Summary
Experienced Technical Manager with 8 years in managing and leading high-performance teams. Proven track record in project management, cost reduction, and operational efficiency. Expertise in Agile methodologies, software development, and process improvement.
Work History
Technical Manager
InnovateTech Solutions - Springfield, IL
June 2024 - March 2026
- Led team to achieve 25% increase in project completion rate
- Managed 2M budget, reduced costs by 15%
- Implemented Agile, improved efficiency by 20%
Senior Technical Lead
TechDrive Inc. - Springfield, IL
January 2021 - May 2024
- Oversaw deployment of 10+ products globally
- Enhanced system uptime by 30%
- Directed 15-member team across multiple projects
Software Engineer
CodeMinds Corp - Chicago, IL
March 2018 - December 2020
- Developed 50+ software solutions to clients
- Cut development time by 40% using new technologies
- Trained junior engineers, enhanced team competency
Languages
- Spanish - Beginner (A1)
- French - Intermediate (B1)
- German - Beginner (A1)
Skills
- Project Management
- Agile Methodologies
- Software Development
- Team Leadership
- Budget Management
- Process Improvement
- Risk Management
- Technical Documentation
Certifications
- PMP Certification - PMI
- Certified ScrumMaster - Scrum Alliance
Education
Master's in Computer Science Software Engineering
Stanford University Stanford, California
December 2017
Bachelor's in Computer Science Computer Science
University of California, Berkeley Berkeley, California
May 2014
How to Write a Technical Manager Resume Summary
Your resume summary is the first chance to captivate hiring managers, making it important for setting a positive tone. As a technical manager, you should emphasize your leadership abilities and technical expertise that align with the role’s demands. To illustrate what makes an effective summary, we will look at examples that highlight strong qualifications and common pitfalls to avoid:
I am a technical manager with extensive experience in various technologies. I seek a challenging position where I can use my knowledge and lead projects effectively. A company that values innovation and offers career advancement opportunities is what I'm looking for. I believe my background will help the team achieve great results.
- Lacks specific details about technical skills and leadership experience, making it too vague
- Overuses personal pronouns which detracts from professionalism
- Emphasizes personal desires instead of clearly articulating what value the applicant brings to potential employers
Results-driven technical manager with over 8 years of experience in leading cross-functional teams to deliver innovative software solutions. Successfully improved project delivery timelines by 20% while reducing costs by 15% through the implementation of Agile methodologies and process optimization. Proficient in cloud technologies, DevOps practices, and strategic planning to align technology initiatives with business goals.
- Starts with a clear indication of experience level and managerial focus
- Highlights quantifiable achievements that reflect both efficiency improvements and cost savings
- Showcases relevant technical skills that are essential for a technical management role
Pro Tip
Showcasing Your Work Experience
The work experience section is important for your resume as a technical manager, representing the bulk of your content. Good resume templates prominently feature this section to highlight your relevant experience.
This part of your resume should be organized in reverse-chronological order, detailing your previous roles. Use bullet points to summarize key achievements and contributions in each position, showcasing your leadership and technical skills.
To further assist you, we will provide examples that illustrate strong work history entries for technical managers. These examples will clarify what makes an entry stand out and what pitfalls to avoid.
Technical Manager
Tech Solutions Inc. – San Francisco, CA
- Oversaw technical projects.
- Managed a team of engineers.
- Communicated with clients.
- Ensured project goals were met.
- Lacks specific employment dates to provide context
- Bullet points are too vague and do not highlight specific achievements or skills
- Focuses on general responsibilities rather than measurable outcomes that demonstrate effectiveness
Technical Manager
Tech Innovations Inc. – San Francisco, CA
March 2020 - Current
- Lead a team of software engineers to develop cutting-edge applications, increasing product delivery speed by 30%.
- Implement agile methodologies that improve project efficiency, contributing to a 40% reduction in time-to-market for new features.
- Facilitate cross-departmental workshops to foster collaboration and innovation, resulting in the launch of three successful products within one year.
- Each bullet begins with powerful action verbs that clearly articulate the applicant’s contributions
- Incorporates specific metrics showing quantifiable success in performance improvements
- Showcases relevant managerial skills and leadership qualities essential for the role
While your resume summary and work experience are important elements, don't overlook the importance of other sections that contribute to a well-rounded application. Each part plays a role in showcasing your qualifications. For more insights on crafting an effective resume, explore our complete guide on how to write a resume.
Top Skills to Include on Your Resume
A well-structured skills section is a critical component of any effective resume. It provides immediate clarity for employers, allowing them to quickly identify the qualifications that align with their needs.
This section enables hiring managers to efficiently gauge applicant suitability for the role while empowering applicants to highlight how their expertise meets job requirements. Technical manager professionals should emphasize both technical and interpersonal skills, which will be elaborated on in the following sections.
For technical managers, soft skills are essential because they facilitate communication, teamwork, and problem-solving. These skills ultimately drive project success and foster a positive work environment.
When selecting skills for your resume, it’s important to align them with what employers expect from applicants. Many organizations use automated systems to filter out applications that lack essential resume skills, making a tailored approach important.
To improve your chances, thoroughly review job postings for insights on which specific resume skills are in demand. This strategy helps ensure that you highlight the qualifications that will engage both recruiters and ATS directly.
Pro Tip
10 skills that appear on successful technical manager resumes
Highlighting essential skills in your resume can capture the attention of recruiters looking for technical managers. You can see these skills effectively demonstrated in our resume examples, giving you the advantage of a polished and professional application.
By the way, incorporating relevant skills that align with your expertise will improve your chances of landing an interview. Consider including the following ten skills in your resume if they match your qualifications:
Project management
Team leadership
Technical problem-solving
Agile methodologies
Software development life cycle (SDLC)
Data analysis
Budget management
Vendor relations
Risk assessment
Quality assurance
Based on analysis of 5,000+ computer software professional resumes from 2023-2024
Resume Format Examples
Choosing the right resume format is important for a technical manager, as it highlights your key skills, relevant experience, and career advancements in a clear and strong way.
Functional
Focuses on skills rather than previous jobs
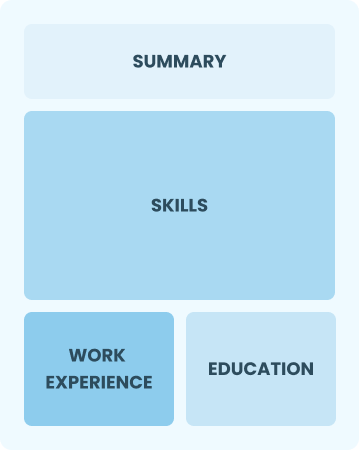
Best for:
Recent graduates and career changers with up to two years of experience
Combination
Balances skills and work history equally
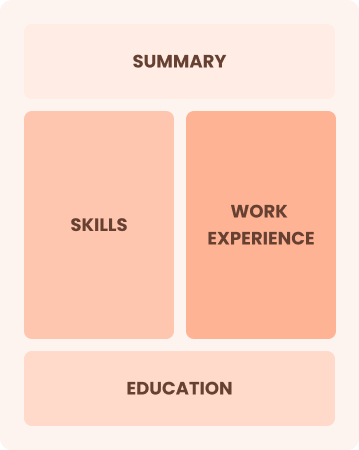
Best for:
Mid-career professionals eager to demonstrate their skills and pursue new opportunities
Chronological
Emphasizes work history in reverse order

Best for:
Leaders with extensive expertise in technical project management
Frequently Asked Questions
Should I include a cover letter with my technical manager resume?
Absolutely, including a cover letter can significantly improve your application by showcasing your unique skills and enthusiasm for the role. It allows you to elaborate on your experiences and connect them to the job you're applying for. For easy assistance, check out our comprehensive guide on how to write a cover letter or use our Cover Letter Generator for quick results.
Can I use a resume if I’m applying internationally, or do I need a CV?
When applying for jobs abroad, a CV is often required instead of a resume. This is especially true in Europe and other regions. For guidance on how to write a CV, explore our resources that provide CV examples and tips tailored to international standards.
What soft skills are important for technical managers?
Soft skills like leadership, problem-solving, and interpersonal skills are important for a technical manager. These skills help you guide teams, resolve conflicts, and foster collaboration. By honing these abilities, you can ensure projects progress smoothly while maintaining strong professional relationships.
I’m transitioning from another field. How should I highlight my experience?
Highlight your transferable skills such as project management, team leadership, and effective communication. Even if your past roles are outside the tech industry, these abilities are important for a technical manager. Provide specific examples that align your achievements with the responsibilities of the position to showcase your readiness to excel in this new role.
Where can I find inspiration for writing my cover letter as a technical manager?
For those pursuing technical manager roles, exploring professionally crafted cover letter examples can be invaluable. These samples offer inspiration for content ideas, formatting styles, and effective ways to showcase your skills and experiences, helping you stand out in a competitive job market.
How should I format a cover letter for a technical manager job?
To format a cover letter, begin with your name and contact details. Follow this with a warm greeting, an engaging introduction that highlights your interest in the technical manager role, and a concise overview of your skills. Remember to customize each letter to align with the job description. Conclude by inviting further discussion about your application.







