Hard skills include abilities such as inventory management, shipment tracking, and quality control inspections that are essential for shipping and receiving clerks to ensure efficiency.
Popular Shipping And Receiving Clerk Resume Examples
Check out our top shipping and receiving clerk resume examples, showcasing key skills such as inventory management, order processing, and strong attention to detail. These samples will help you communicate your strengths to potential employers.
Are you ready to build your own impressive resume? Our Resume Builder offers easy-to-use templates tailored for logistics professionals, making the process straightforward and efficient.
Recommended
Entry-level shipping and receiving clerk resume
This entry-level resume for a shipping and receiving clerk highlights the job seeker's logistics skills and achievements in optimizing warehouse operations, including improving accuracy and efficiency. New professionals in this field must showcase their ability to manage processes and drive productivity, even with limited direct experience.
Mid-career shipping and receiving clerk resume
This resume effectively emphasizes the applicant's logistics expertise and leadership skills, showcasing a proven ability to improve efficiency and accuracy. Their accomplishments reflect readiness for greater challenges, demonstrating a trajectory toward more complex roles in shipping and receiving operations.
Experienced shipping and receiving clerk resume
This resume highlights the applicant's robust experience in logistics, underscoring achievements such as processing over 1,000 shipments monthly and reducing receiving errors by 15%. This clear formatting improves readability, making it ideal for employers seeking efficient applicants.
Resume Template—Easy to Copy & Paste
Tao Wang
Portland, ME 04112
(555)555-5555
Tao.Wang@example.com
Skills
- Inventory Management
- Logistics Coordination
- Shipping Solutions
- Supply Chain Optimization
- Warehouse Operations
- Database Management
- Cost Reduction Strategies
- Shipping Compliance
Certifications
- Certified Logistics Technician - Manufacturing Skill Standards Council
- Certified Supply Chain Professional - APICS
- Forklift Safety Certification - National Safety Council
Languages
- Spanish - Beginner (A1)
- French - Beginner (A1)
- German - Beginner (A1)
Professional Summary
Detail-oriented Shipping and Receiving Clerk with six years of robust experience in logistics coordination and inventory management. Proven track record in optimizing supply chain efficiency and reducing costs. Skilled in data management and compliance with safety standards.
Work History
Shipping and Receiving Clerk
CargoMaster Logistics - Portland, ME
March 2024 - March 2026
- Managed inventory levels, reducing errors by 30%
- Coordinated daily shipments and deliveries efficiently
- Improved packaging processes, cutting costs by 15%
Inventory Coordinator
Warehouse Solutions - Portland, ME
March 2021 - February 2024
- Analyzed inventory trends, boosting turnover rate by 25%
- Maintained accurate records of shipments and receipts
- Implemented database improvements for tracking items
Logistics Assistant
Efficient Transport Corp - Westbrook, ME
March 2020 - February 2021
- Assisted in transport coordination and scheduling
- Optimized routes saving 10% on delivery times
- Ensured compliance with safety and quality standards
Education
Master of Business Administration Supply Chain Management
University of Illinois Urbana-Champaign, Illinois
May 2019
Bachelor of Science Logistics and Transportation
Illinois State University Normal, Illinois
May 2017
How to Write a Shipping And Receiving Clerk Resume Summary
Your resume summary is the first opportunity to make a lasting impression on hiring managers, so it's important that it effectively showcases your qualifications. As a shipping and receiving clerk, you should highlight your organizational skills, attention to detail, and ability to manage inventory efficiently. To illustrate what makes a compelling resume summary for this role, we’ll provide examples that demonstrate best practices and common pitfalls:
I am an experienced shipping and receiving clerk seeking a position that will allow me to use my skills effectively. I have a strong background in logistics and inventory management, and I believe I can help the company succeed. A job that provides good benefits and the chance for advancement is very important to me.
- Contains vague language about skills without providing specific examples of past achievements
- Emphasizes personal desires rather than demonstrating how the applicant can add value to the employer's operations
- Uses generic phrases that fail to highlight unique qualifications or experiences relevant to shipping and receiving
Detail-oriented shipping and receiving clerk with over 7 years of experience in logistics management and inventory control. Successfully improved shipping accuracy by 20% through the implementation of a new barcode scanning system, resulting in decreased order errors. Proficient in using warehouse management software and operating forklifts, ensuring efficient material handling and timely deliveries.
- Begins with clear years of experience and specific job functions within logistics
- Highlights a quantifiable achievement that showcases a significant improvement in operational efficiency
- Mentions relevant technical skills that are essential for success in shipping and receiving roles
Pro Tip
Showcasing Your Work Experience
The work experience section is important for your resume as a shipping and receiving clerk, where you'll showcase the bulk of your content. Effective resume templates always prioritize this section to highlight your relevant experience.
Organize this part in reverse-chronological order, listing your previous positions clearly. Use bullet points to detail your key achievements and responsibilities that demonstrate your skills in shipping and receiving operations.
To help you understand what makes a strong work history entry, we’ve prepared a couple of examples. These examples will reveal what works well and what pitfalls to avoid:
Shipping and Receiving Clerk
ABC Logistics – Houston, TX
- Received shipments and checked items.
- Organized storage areas.
- Used a computer for tracking inventory.
- Assisted in loading and unloading trucks.
- Lacks specific details about accomplishments or metrics
- Bullet points are overly simplistic and do not highlight skills or strengths
- Focuses on routine tasks rather than achievements or contributions to efficiency
Shipping and Receiving Clerk
ABC Logistics – Los Angeles, CA
March 2020 - Present
- Oversee receiving and inspection of shipments, ensuring accuracy with a 98% error-free rate.
- Coordinate the timely dispatch of outgoing orders, achieving a 15% reduction in delivery times through efficient routing.
- Maintain inventory records using warehouse management software, contributing to a 20% decrease in stock discrepancies.
- Starts each bullet with strong action verbs that clearly show the job seeker's contributions
- Incorporates specific metrics to highlight effectiveness and impact on operations
- Demonstrates relevant skills such as attention to detail and organizational abilities tied to job responsibilities
While the resume summary and work experience are important components of your resume, don’t overlook the importance of other sections. Each part plays a role in showcasing your qualifications. For more detailed insights, be sure to check out our comprehensive guide on how to write a resume.
Top Skills to Include on Your Resume
A skills section is vital for any resume as it provides a snapshot of the applicant's capabilities, helping both job seekers and employers focus on essential qualifications. This concise part of the resume highlights relevant abilities that align with job requirements, making it easier for hiring managers to identify potential fits.
This section allows employers to swiftly evaluate whether job seekers possess necessary competencies before reviewing detailed experiences. Shipping and receiving clerks should emphasize both technical skills and organizational abilities, which will be elaborated further below.
Soft skills are essential for shipping and receiving clerks, as strong communication and teamwork improve efficiency, accuracy, and overall workplace harmony in logistics operations.
Selecting the right resume skills is important, as it aligns your qualifications with what employers expect from ideal job seekers. Many organizations use automated systems that filter out applicants who lack essential skills for the position.
To improve your chances of getting noticed, carefully review job postings to identify which skills are emphasized. This approach not only appeals to recruiters but also ensures your resume is optimized for ATS, increasing your likelihood of advancing in the hiring process.
Pro Tip
10 skills that appear on successful shipping and receiving clerk resumes
Showcasing in-demand skills on your resume can significantly improve your attractiveness to potential employers. By including these skills, you demonstrate that you're well-equipped for shipping and receiving clerk positions. You can see these competencies illustrated in our resume examples, which will empower you to apply with confidence.
By the way, consider incorporating relevant skills from this list when they align with your experience and job criteria:
Attention to detail
Inventory management
Time management
Communication skills
Problem-solving
Forklift operation
Basic computer skill
Organizational abilities
Teamwork
Safety awareness
Based on analysis of 5,000+ administrative professional resumes from 2023-2024
Resume Format Examples
Choosing the right resume format is important for a shipping and receiving clerk, as it showcases your logistical skills, relevant experience, and career advancement in an organized manner.
Functional
Focuses on skills rather than previous jobs
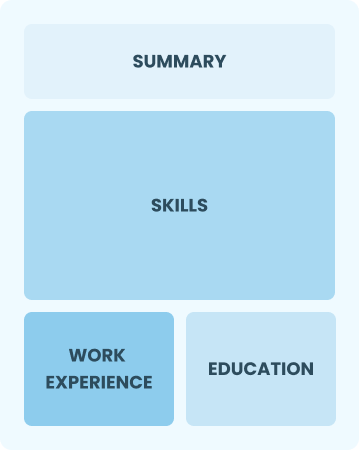
Best for:
Recent graduates and career changers with little to no experience in logistics
Combination
Balances skills and work history equally
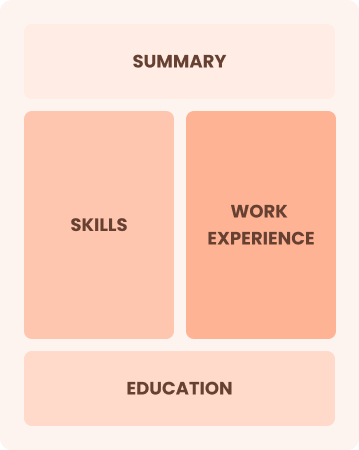
Best for:
Mid-career professionals focused on demonstrating skills and pursuing new opportunities
Chronological
Emphasizes work history in reverse order

Best for:
Seasoned clerks excelling in logistics and team leadership
Frequently Asked Questions
Should I include a cover letter with my shipping and receiving clerk resume?
Absolutely, including a cover letter can significantly improve your application by showcasing your personality and explaining how your skills align with the job. It offers a chance to make a memorable first impression. To get started, explore our comprehensive guide on how to write a cover letter or use our Cover Letter Generator for a quick and easy solution.
Can I use a resume if I’m applying internationally, or do I need a CV?
When applying for jobs outside the U.S., use a CV instead of a resume. A CV provides a comprehensive overview of your academic and professional history, ideal for international roles. Explore our resources on how to write a CV and look at some CV examples to ensure your application stands out globally.
What soft skills are important for shipping and receiving clerks?
Soft skills such as attention to detail, teamwork, and interpersonal skills are essential for a shipping and receiving clerk. These abilities help foster collaboration with colleagues and ensure accuracy in inventory management, which ultimately improves operational efficiency and builds trust within the team.
I’m transitioning from another field. How should I highlight my experience?
When applying for shipping and receiving clerk positions, highlight your transferable skills like organization, attention to detail, and communication. These abilities demonstrate your potential value, even if you lack direct experience. Use specific examples from past roles to illustrate how you've managed tasks similar to those in this field.
Where can I find inspiration for writing my cover letter as a shipping and receiving clerk?
For individuals seeking shipping and receiving clerk positions, exploring professionally crafted cover letter examples can be invaluable. These samples offer inspiration for content ideas, provide formatting guidance, and help you showcase your qualifications to stand out to potential employers. Use them to elevate your application materials and make a strong impression.
Should I include a personal mission statement on my shipping and receiving clerk resume?
Including a personal mission statement on your resume is advisable. It conveys your values and career aspirations, particularly to companies that prioritize strong organizational cultures or community-focused missions. This approach can set you apart in your job search.







