Hard skills like data analysis, process auditing, and quality control testing are essential for a quality assurance specialist to ensure product excellence.
Popular Quality Assurance Specialist Resume Examples
Check out our top quality assurance specialist resume examples that highlight critical skills such as attention to detail, process improvement, and compliance. These examples will help you showcase your qualifications and achievements to potential employers.
Ready to build your impressive resume? Our Resume Builder offers user-friendly templates specifically designed for quality assurance professionals, making it simple to create a standout application.
Recommended
Entry-level quality assurance specialist resume
This entry-level resume highlights the job seeker's expertise in quality assurance through quantifiable achievements, showcasing their ability to reduce defect rates and improve efficiency within teams. New professionals in this field should demonstrate their technical skills and relevant certifications, illustrating their readiness to contribute despite limited experience.
Mid-career quality assurance specialist resume
This resume effectively showcases the applicant's extensive experience and accomplishments in quality assurance, emphasizing their readiness for leadership roles. The clear presentation of metrics-driven achievements illustrates a solid career progression and capability to tackle complex challenges in QA management.
Experienced quality assurance specialist resume
This resume highlights the applicant's extensive experience in quality assurance, showcasing achievements such as improving software testing efficiency by 40% and reducing defect rates by 25%. The clear bullet points improve readability, allowing hiring managers to quickly assess key accomplishments.
Resume Template—Easy to Copy & Paste
Chris Chen
Chicago, IL 60604
(555)555-5555
chris@example.com
Professional Summary
Proven QA specialist enhancing processes by 30%, skilled in ISO compliance, producing zero-defect deliverables using advanced methodologies in dynamic software environments.
Work History
Quality Assurance Specialist
Precision Tech Solutions - Chicago, IL
April 2025 - April 2026
- Enhanced QA processes by 30%
- Reduced defects by 15% using testing tools
- Led audits ensuring 100% compliance
Quality Analyst
DataWave Analytics - Chicago, IL
April 2023 - March 2025
- Increased test coverage by 20%
- Implemented testing, lowering errors by 10%
- Collaborated in team for ISO standards
QA Tester
SysLogic Software - Springfield, IL
April 2022 - March 2023
- Reduced testing time by 25%
- Executed tests identifying 300+ bugs
- Developed optimized scripts improving efficiency
Languages
- Spanish - B1
- Intermediate - French
- B1 - Intermediate
- Mandarin - B1
- Intermediate -
Skills
- Quality Control
- Regression Testing
- Automated Testing
- Agile Methodologies
- Defect Tracking
- Test Plan Development
- ISO 9001 Compliance
- Data Analysis
Certifications
- Certified Software Quality Analyst - Quality Assurance Institute
- Agile Tester Certification - International Software Testing Qualifications Board
Education
Master's Degree Software Quality Management
Harvard University Springfield, IL
May 2021
Bachelor's Degree Computer Science
Boston University Springfield, IL
June 2019
How to Write a Quality Assurance Specialist Resume Summary
Your resume summary is the first opportunity to catch an employer's attention, so it's essential that you make it count. As a quality assurance specialist, you should emphasize your skills in identifying issues and ensuring standards are met to assure employers of your capabilities. To guide you in crafting this critical section, we will provide examples that illustrate effective approaches and common pitfalls to avoid:
I am a dedicated quality assurance specialist with many years of experience in the field. I am seeking a position where I can leverage my skills to contribute to the success of the company. A supportive work environment that allows me to develop professionally is what I hope for. I believe I can add value to your team if given a chance.
- Lacks specific details about qualifications and achievements, making it less strong
- Relies heavily on personal statements, which dilute professional intent
- Emphasizes personal desires instead of highlighting how the job seeker can meet the employer's needs
Detail-oriented quality assurance specialist with over 7 years of experience in the pharmaceutical industry, focusing on compliance and process improvement. Achieved a 20% reduction in product defects through rigorous testing protocols and quality audits. Proficient in statistical analysis software, root cause analysis, and regulatory compliance to ensure adherence to GMP standards.
- Starts with precise experience level and industry focus, establishing credibility
- Features quantifiable achievements that highlight the impact on product quality
- Includes specific technical skills relevant to quality assurance roles that are valued by employers
Pro Tip
Showcasing Your Work Experience
The work experience section is important for your resume as a quality assurance specialist, serving as the main focus where you’ll present the bulk of your content. Good resume templates always prioritize this section to highlight your professional journey.
This part should be organized in reverse-chronological order, listing your previous roles clearly. Use bullet points to detail your key achievements and contributions in each position, ensuring they reflect your expertise in quality assurance.
To illustrate effective work history entries for quality assurance specialists, we’ve prepared some examples that show what makes an impact and what pitfalls to avoid:
Quality Assurance Specialist
Tech Innovations Inc. – San Francisco, CA
- Tested software products.
- Documented results and findings.
- Collaborated with development teams.
- Ensured compliance with quality standards.
- Lacks specific employment dates to establish timeline
- Bullet points are vague and do not highlight achievements or impact
- Focuses on routine tasks rather than measurable contributions to product quality
Quality Assurance Specialist
Tech Innovations Inc. – San Francisco, CA
March 2020 - Present
- Develop and execute comprehensive test plans for software applications, ensuring compliance with industry standards and reducing bug reports by 30%.
- Collaborate with development teams to identify and resolve issues early in the process, leading to a 20% decrease in project delays.
- Train new QA staff on testing methodologies and tools, improving team efficiency and maintaining high-quality deliverables.
- Starts each bullet with compelling action verbs that convey the applicant's role in achieving outcomes
- Incorporates quantifiable metrics to highlight the effectiveness of their contributions
- Demonstrates relevant skills that align with quality assurance requirements
While your resume summary and work experience sections are important, don’t overlook other significant areas. Each section contributes to your overall presentation. For detailed guidance on optimizing every part of your resume, please refer to our comprehensive guide on how to write a resume.
Top Skills to Include on Your Resume
Including a skills section on your resume is important as it provides a clear snapshot of your qualifications. This segment not only showcases your capabilities but also helps you stand out to potential employers seeking specific expertise.
Hiring managers appreciate this organized format, allowing them to quickly evaluate whether applicants align with their needs. For quality assurance specialists, it's vital to highlight both technical and interpersonal skills, which will be discussed in further detail below.
Soft skills are essential for quality assurance specialists, as they improve communication, teamwork, and problem-solving abilities. These skills ensure successful project outcomes and foster a positive work environment.
When selecting skills for your resume, aligning them with what potential employers expect is important. Many organizations use automated screening systems to filter out applicants who lack essential resume skills, making it important to ensure a match.
To highlight your qualifications, take the time to review job postings relevant to your field. These listings offer valuable insights into which skills are most sought after by recruiters and can help you tailor your resume for both human eyes and ATS scanning.
Pro Tip
10 skills that appear on successful quality assurance specialist resumes
To grab the attention of hiring managers, it's essential to highlight key skills that are highly sought after in quality assurance specialist roles. Showcasing these critical competencies in your resume examples can provide the confidence needed to apply for positions.
By the way, consider adding these relevant skills to your resume if they align with your experience and job requirements:
Attention to detail
Analytical thinking
Problem-solving
Communication skills
Team collaboration
Knowledge of testing methodologies
Familiarity with automated testing tools
Understanding of software development life cycle (SDLC)
Time management
Quality control standards
Based on analysis of 5,000+ sciences professional resumes from 2023-2024
Resume Format Examples
Choosing the right resume format is important for a quality assurance specialist, as it showcases essential skills, relevant experience, and career growth in the field.
Functional
Focuses on skills rather than previous jobs
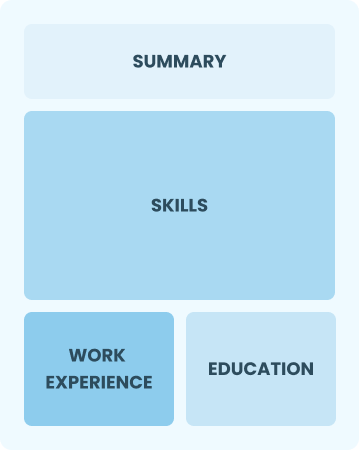
Best for:
Recent graduates and career changers with up to two years of experience
Combination
Balances skills and work history equally
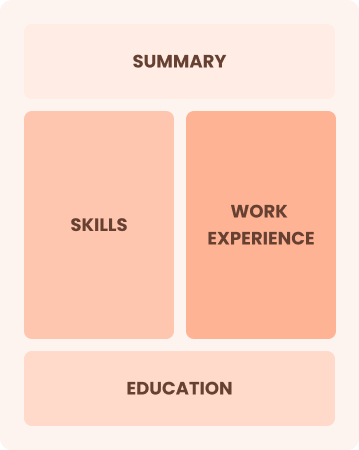
Best for:
Professionals showcasing quality expertise and career progression
Chronological
Emphasizes work history in reverse order

Best for:
Experts leading quality initiatives and improving compliance standards
Frequently Asked Questions
Should I include a cover letter with my quality assurance specialist resume?
Absolutely, including a cover letter can significantly improve your application by showcasing your passion and qualifications. It allows you to personalize your story and make a memorable impression on hiring managers. If you're looking for guidance, take a look at our tips on how to write a cover letter or use our Cover Letter Generator for quick assistance.
Can I use a resume if I’m applying internationally, or do I need a CV?
When applying for jobs abroad, a CV is often preferred over a resume. This is especially true in Europe and Asia where detailed academic and professional histories are valued. Explore our resources for CV examples and tips on how to write a CV to ensure your application meets international expectations.
What soft skills are important for quality assurance specialists?
Soft skills such as attention to detail, critical thinking, and interpersonal skills in effective communication are essential for quality assurance specialists. These abilities foster collaboration with teams, improve problem-solving, and ensure that products meet standards while building trust with colleagues and clients alike.
I’m transitioning from another field. How should I highlight my experience?
Highlight your transferable skills like attention to detail, analytical thinking, and effective communication. These traits are key for a quality assurance specialist and can be applied from diverse past roles. Incorporate examples that demonstrate how your earlier accomplishments align with quality assurance responsibilities to emphasize your value in this new field.
How should I format a cover letter for a quality assurance specialist job?
To format a cover letter, start with your name and contact details. Next, include a professional salutation and an engaging introduction about yourself. Highlight skills relevant to the job requirements throughout the letter. Conclude with a strong closing statement that invites further communication. Customizing your letter for each quality assurance specialist position is important to stand out to potential employers.
How do I add my resume to LinkedIn?
To improve your professional visibility, you should add your resume to LinkedIn. Upload it directly or highlight key achievements in the "About" and "Experience" sections. This approach increases the chances of healthcare recruiters and hiring managers finding you among qualified applicants in your field.








